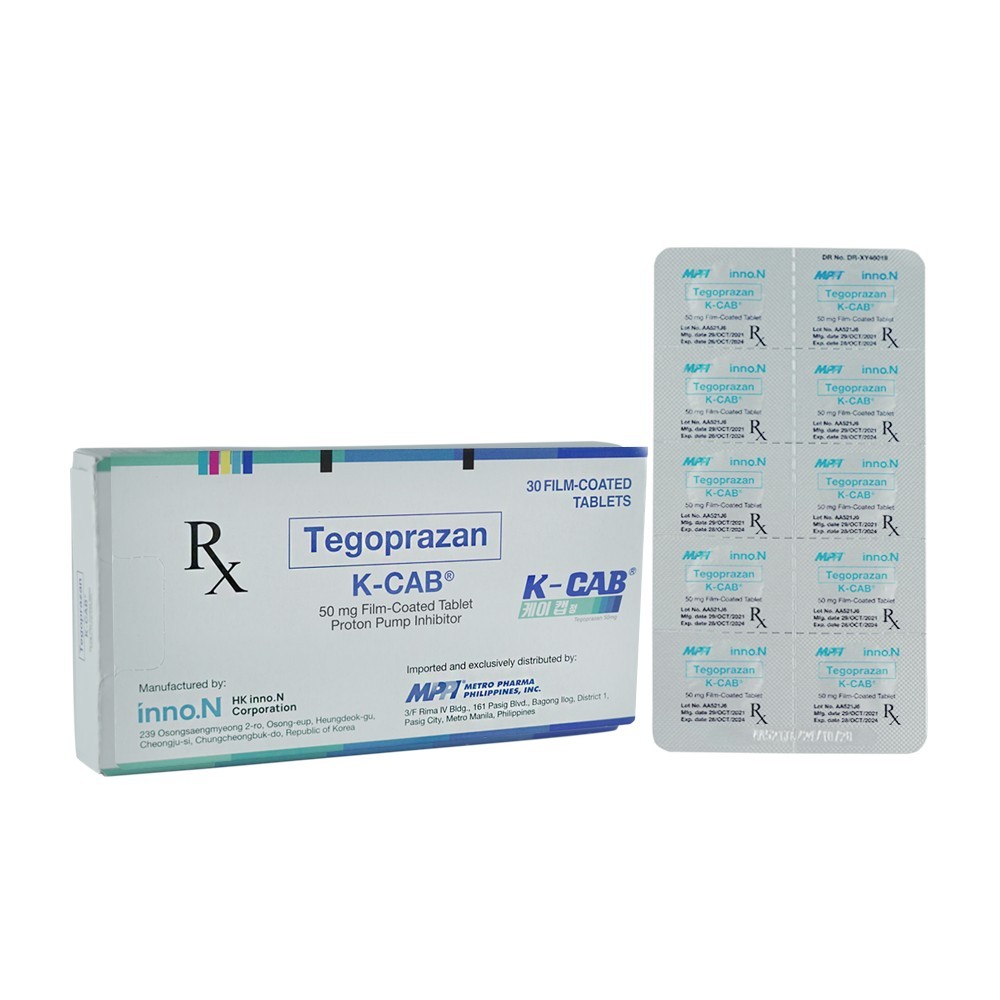
K-CAB 50mg Tablet
GENERIC NAME:
Tegoprazan
FORM:
Film-Coated Tablet
INDICATION AND USAGE:
For treatment of Erosive Gastroesophageal Reflux Disease, Non-Erosive Gastroesophageal Reflux Disease, and Gastric Ulcer.
Eradication of H.pylori concurrently given with appropriate antibiotic therapy treatment in patients with peptic ulcer and/or chronic atrophic gastritis.
DOSAGE AND ADMINISTRATION:
Adult Population:
Treatment of Erosive Gastroesophageal Reflux Disease
- 50 mg once daily for 4 weeks.
- For patients who do not heal or have persistent symptoms after 4 weeks, additional 4-week treatment may be considered.
Treatment of Non-Erosive Gastroesophageal Reflux Disease
- 50 mg once daily for 4 weeks.
Treatment of Gastric Ulcer
- 50 mg once daily for 8 weeks.
Eradication of H.pylori concurrently given with appropriate antibiotic therapy treatment in patients with peptic ulcer and/or chronic atrophic gastritis.
- Patients with H.pylori infection should be treated with eradication therapy. Tegoprazan 50mg, clarithromycin 500mg and amoxicillin 1g are orally administered twice daily for 7 days.
K-CAB can be taken without regard to food.
AVAILABILITY:
Box of 30 Film-Coated Tablets